Sweating Rate and Sweat Sodium Concentration in Athletes: A Review of Methodology and Intra/Interindividual Variability
In this blog, we will be reviewing and summarizing the work that Lindsay B. Baker did on analyzing sweating rate and sweat sodium concentration in athletes. Lindsay worked on this detailed review and we will do a quick deep dive into it and the data presented. Here is the link to the scientific paper review: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5371639/
Sweating is an essential mechanism for regulating body temperature during exercise, and athletes can lose significant amounts of water and electrolytes through sweating. Measuring sweating rate (SR) and sweat electrolyte concentrations can help to guide personalized fluid and electrolyte replacement strategies, but there are many factors that can impact the accuracy of sweat testing. In a recent review paper published in Sports Medicine, researchers examined the literature on the effects of laboratory and field sweat-testing methodological variations on SR and sweat composition, as well as factors that may impact intra/interindividual variability in SR and sweat electrolyte concentrations during exercise.
Accurately measuring SR is crucial for understanding an athlete’s hydration status, and the simplest and most accurate method is to track changes in body mass during exercise. However, there are potential confounding factors to consider, such as non-sweat sources of mass change and trapped sweat in clothing. In addition, the type of collection system used, the timing and duration of sweat collection, skin cleaning procedures, sample storage and handling, and analytical techniques can all affect the measurement of SR and sweat electrolyte concentrations.
One key source of variability in SR and sweat electrolyte concentrations is the type of collection system used. Whole-body collection systems are typically considered to be more accurate, but they can be impractical in some situations, such as during competition. Localized collection systems, which collect sweat from a specific anatomical site, can be more convenient, but they may not provide an accurate representation of whole-body SR and electrolyte losses.
The timing and duration of sweat collection can also impact the accuracy of measurements. Sweat rate typically peaks within the first 30-60 minutes of exercise, and collecting sweat samples over a longer period can lead to overestimates of SR and electrolyte losses. In addition, the timing of sweat collection relative to the onset of exercise can affect the accuracy of measurements.
Skin cleaning procedures, sample storage and handling, and analytical techniques can also impact the accuracy of SR and electrolyte measurements. For example, using saline to clean the skin before collecting sweat samples can affect electrolyte concentrations, and improper storage and handling of samples can lead to inaccurate results. In addition, different analytical techniques can produce varying results, and it is important to use standardized methods to ensure the reliability of measurements.
In addition to methodological variations, there are many factors that can impact intra/interindividual variability in SR and sweat electrolyte concentrations during exercise. These include exercise intensity, environmental conditions, heat acclimation, aerobic capacity, body size and composition, the wearing of protective equipment, sex, maturation, aging, diet, and hydration status. For example, exercise intensity can affect SR and electrolyte concentrations, with higher-intensity exercise leading to higher rates of sweating and electrolyte loss. Environmental conditions, such as temperature and humidity, can also impact SR and electrolyte concentrations, with hotter and more humid conditions leading to higher rates of sweating.
Overall, this review highlights the importance of standardized, well-controlled methods in sweat testing to ensure accurate and reliable results. The information gleaned from sweat testing can be used to guide personalized fluid and electrolyte replacement recommendations for athletes, but unstandardized methodological practices and challenging field conditions can produce inconsistent and inaccurate results. By understanding the factors that can impact the accuracy of sweat testing, scientists and practitioners can develop more effective strategies for measuring SR and electrolyte concentrations, and improve the personalized hydration strategies for athletes.
In addition to measuring SR, it is also important to assess the concentration of electrolytes in sweat, particularly sodium. Sodium is the most abundant electrolyte in sweat, and changes in its concentration can impact hydration status and performance. In their review, the researchers examined the literature on the effects of methodological variations on sweat sodium concentration and found that the average values can vary depending on the method used. For example, one study reported an average sweat sodium concentration of 44.1 mmol/L using a whole-body collection system, while another study reported an average concentration of 59.4 mmol/L using a localized collection system.
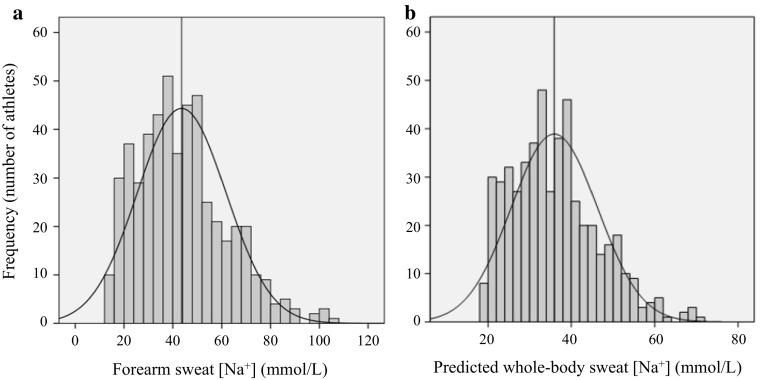
In addition to these average values, the researchers also discussed the distribution of sodium concentrations in sweat, as depicted in a histogram. The histogram showed that there is a wide range of sodium concentrations in sweat, with some individuals having concentrations as low as 20 mmol/L and others as high as 80 mmol/L. This intra/interindividual variability in sodium concentrations highlights the importance of personalized fluid and electrolyte replacement strategies, as a one-size-fits-all approach may not be effective for all athletes.
hDrop provides the data in mg/L of electrolytes since it is easier for the end user to understand. That is why we also wanted to cover the calculations needed to convert mmol/L to mg/L. The concentration of sodium in sweat can be expressed either in millimoles per liter (mmol/L) or in milligrams per liter (mg/L). To convert from mmol/L to mg/L, you need to know the atomic weight of sodium, which is 22.99 g/mol. Since 1 mol of any substance contains Avogadro’s number of atoms (6.022 x 10^23), 1 mmol of a substance contains 6.022 x 10^23/22.99 = 26.05 mg of that substance. Therefore, to convert from mmol/L to mg/L, you need to multiply the concentration in mmol/L by 26.05.
For example, if the average concentration of sodium in sweat is 44.1 mmol/L, the equivalent concentration in mg/L would be 44.1 x 26.05 = 1150.6 mg/L. Similarly, if the concentration of sodium in sweat is 59.4 mmol/L, the equivalent concentration in mg/L would be 59.4 x 26.05 = 1554.1 mg/L. It is worth noting that the conversion from mmol/L to mg/L is only an approximation, as it assumes that the atomic weight of sodium is constant and does not take into account any potential isotope variations. However, for practical purposes, this approximation is generally sufficient for converting between the two units of concentration.
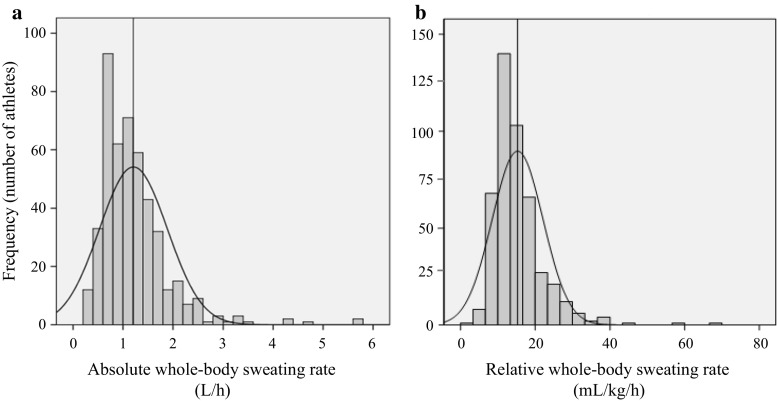
The same is true for SR, as the literature has shown that there is a wide range of SR values among individuals, even when similar exercise protocols are used. For example, one study reported a range of SR values from 0.3 to 2.6 L/h in a group of trained male cyclists, with an average value of 1.3 L/h. Another study reported a range of SR values from 0.5 to 2.5 L/h in a group of trained male triathletes, with an average value of 1.4 L/h. These values demonstrate the need for individualized fluid and electrolyte replacement strategies, as the same recommendations may not be appropriate for all athletes.
Well, we learned quite a lot about hydration through this paper review! hDrop is backed with science and we will keep working on a series of paper review blogs and analyses since we think it helps our customer base understand their fueling strategies. We have hDrop, the first real-time hydration wearable device that does not use sweat disposable patches, and we are committed to keeping bring you the latest news on hydration and athletic performance.
